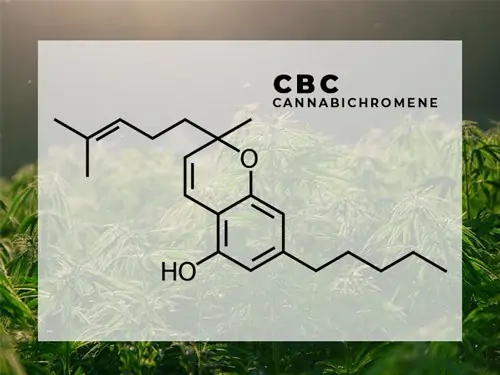
CBC – Cannabichromene
CBC Cannabichromene – Although first discovered in 1966, CBC research, like research on many other cannabinoids, has been held back by federal restrictions. Recently since 2020, the research has been ramping up significantly and has shown the medical potential many cannabinoids have.
Modern breeding practices have focused on CBD and THC, so CBC concentrations have fallen, but CBC has always been one of the Big Six cannabinoids. Like THC and CBD, CBCa is formed from CBGa. CBCa then decarboxylates into CBC. It is mostly found in low concentrations in modern strains of Cannabis and HEMP.
CBC IN PHOENIX • BUY CBC PHOENIX • CBC NEAR ME • CANNABICHROMENE PHOENIX
What We Know About CBC
Despite being one of the most prominent cannabinoids in cannabis, CBC does not get the attention that it deserves. As a non-psychoactive compound, it offers several of the benefits that THC does while not having such pronounced mental effects.
Research has shown that CBC does not interact significantly with CB1 receptors in the body, but that it does have some interactions with the CB2 receptors ( more immune system ). It also interacts with TRPV1 and TRPA1 receptors, both of which are linked to pain, meaning that it can relieve pain with minimal to no psychoactive impact.
Potential Uses for CBC Include:
Anti-Bacterial
- Research as far back as 1981 has found that CBC is especially useful in fighting bacteria. It has substantial effects on a variety of bacteria, including acid-fast strains. It has moderate effects as an anti-fungal as well as an anti-viral.
Anti-Inflammatory and Anti-Pain
- In the current decade, pain is one of the most common reasons people use prescribed substances. CBC appears to be an effective agent at reducing inflammation without directly interacting with cannabinoid receptors. Although some suggest its ability to reduce inflammation is stronger with the presence of other cannabinoids like CBD and CBG. It can reduce swelling and inflammation in the digestive tract. CBC targets pain at the spinal level, and although not as effective at THC at relieving pain, it does so without psychoactive effects.
Anti-Depressant
- Millions of people worldwide suffer from stress and anxiety due to disorders and traumas. When used with other cannabinoids, the research found CBC contributes to even superior mood-elevating effects while activating different pathways in the brain than THC.
Cell Growth
- A 2013 study on mice found that CBC may promote new cell growth. It has also been found to promote the growth of brain cells and to fight migraines. CBC helps the production of astroglial cells, which fight against conditions such as Alzheimer’s disease and dementia.
Anti-Cancer
- A 2006 Study found CBC to be the second most effective cannabinoid at preventing tumor growth. One day CBC could be a powerful chemopreventative agent. It is also shown to inhibit the absorption of anandamide, which is linked to a reduction in a variety of cancers.
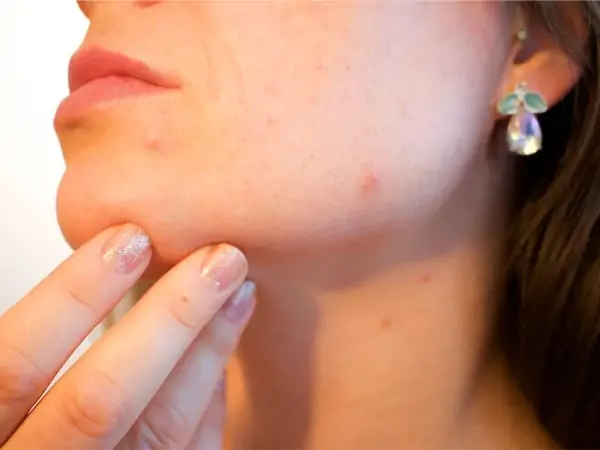
Anti-Acne
- One of the most rampant insecurities people suffer from are caused by common acne. CBC has been found not only to reduce the production of sebum, which is necessary for acne to form, but also to lessen the effects of arachidonic acid. These combined effects prove that CBC could be a potent tool in preventing and managing acne in the future.
Where To Buy CBC In Phoenix
azWHOLEistic offers high quality CBC in Phoenix at our 4 locations, and our online store. Click the button below to view:

 Phoenix
Phoenix Chandler
Chandler Sun City
Sun City Oils & Tinctures
Oils & Tinctures Gummies & Capsules
Gummies & Capsules Topicals
Topicals Inhalation
Inhalation CBD For Pets
CBD For Pets